X5
Find the new EDC/eCRF
Xolomon is a state-of-the-art online platform for the creation of electronic data collection notebooks (EDC/eCRF). The flexibility and adaptability of Xolomon makes it the best solution for data collection in any type of clinical study, from small observational studies to large and complex clinical trials.
Designed for you
The Xolomon platform was developed from the outset by a team of computer engineers, biomedical engineers and healthcare professionals with the aim of providing an optimal solution.
We offer an agile and efficient response to the needs of data capture and management in clinical trials. We also comply with the most demanding data protection standards and applicable regulations (European legislation, such as FDA, GDPR).
Our experience of more than ten years in the development of EDCs for all types of studies, therapeutic areas and clients allows us to provide significant added value to product development.
EDC X5 allows you to create, in a personalised way, different types of roles, assigning access, viewing, editing and exporting permissions, depending on each type of user
Creation of customised portals for the organisation or studio, with a content management system for uploading documentation or specific information
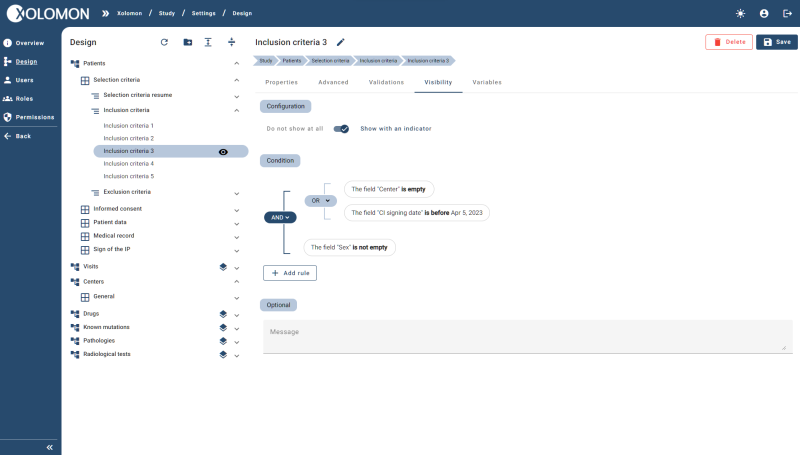
Design your own notebook with X5 technology and its Design section. X5 allows users to build their EDC from scratch without the help of technical profiles
Design and Do It Yourself licence gives you the power to you decide the behaviour, include or remove functionalities and manage the permissions, as well as others
Xolomon is an agile tool that allows you to create EDCs quickly
EDC Xolomon features a fast and intuitive web interface, enhanced by X5
Ability to manage all types of data, including files, conventional and DICOM images
With Xolomon, it is very easy and quick to make necessary changes
EDC X5 includes a forms’ library to allow users to save configured entities for reuse from one study to the next
Real-time data validation
Visibility rules: dynamic forms, with display depending on the information entered
EDC Xolomon has computed fields for the calculation of formulas related to variables or scales
Daily backups
All data is transmitted and stored encrypted with access via HTTPS and SSL communications.
Xolomon is hosted in the cloud, in Microsoft Azure on servers within the EU
Customisable user roles that, together with user groups, provide a powerful security model for controlling access to the application
Xolomon complies with European GDPR, EMA, and FDA 21 part 11 regulations
Xolomon is based on Microsoft technology
With Xolomon you can create EDC for all types of studies, from observational to large clinical trials
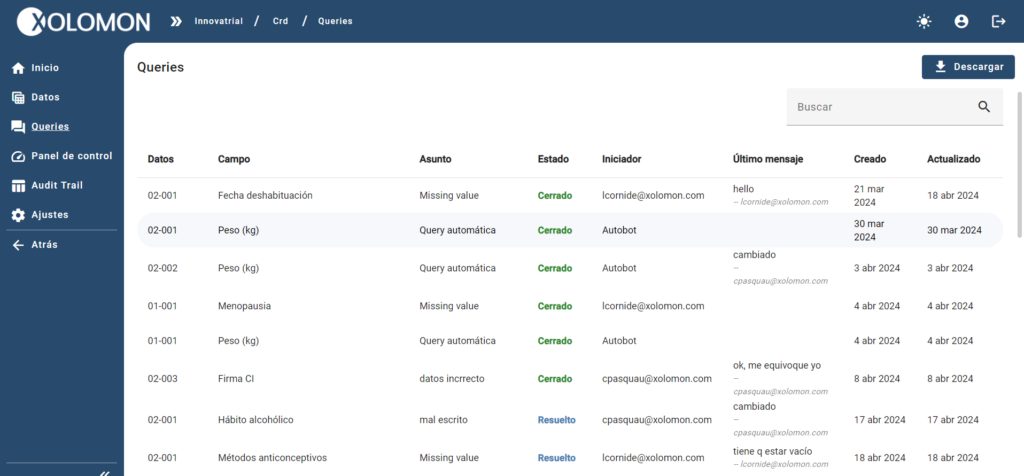
Xolomon implements a powerful system of validations, queries (manual and automatic) and traceability through Audit Trail, which allows you to collect data with the highest quality
Export of data in the main formats for subsequent statistical exploitation
Xolomon offers the possibility of creating customised reports to monitor the evolution of the study in real time
EDC Xolomon is a multi-centric tool, accessible from different locations.
Xolomon offers significant cost savings due to the rapid development of data collection notebooks
As it is SaaS (Software as a Service), there is no need to install software or invest in servers
Xolomon offers a variety of flexible licensing models
Possibility to access from any browser, including older versions
Integrate your study’s ePROs with X5 from within the EDC editor
Incorporate forms, collect data, closely attend to patients and much more with ePROs X5

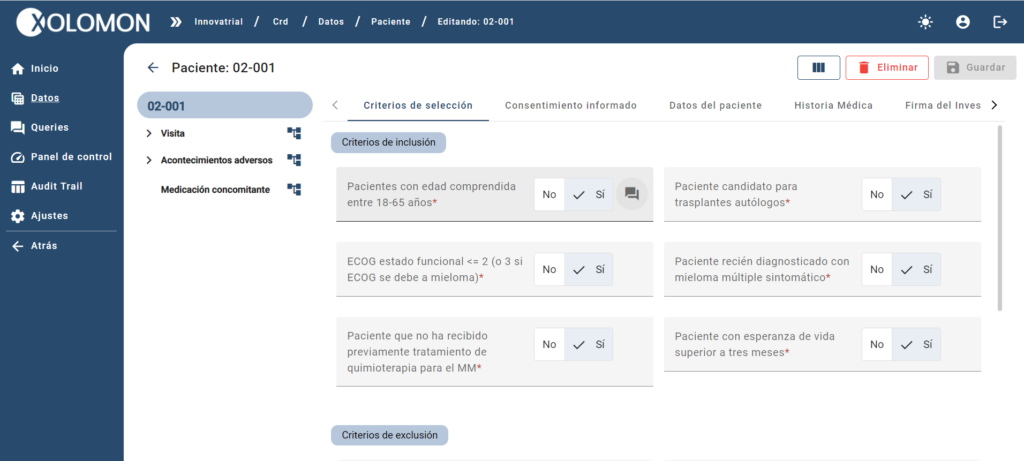
X5 implements a powerful system of validations, queries (manual and automatic) and traceability through Audit Trail, which allows you to collect data with the highest quality
X5 offers a high level of customisation of eCRF
Are you interested in this product? Request more information

